
Chemistry, 09.07.2019 01:00 saabrrinnaaa
Consider the following equilibrium: 4fe(s) + 3 o2(g) < -- --> 2fe2o3(s); which of the following equations is wrong? kp=kc(rt)-5kp= kc(rt)-3kp = po2-3kc=[o2]-3

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, nmadrigal
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 21.06.2019 21:30, sbhishop19
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1
You know the right answer?
Consider the following equilibrium: 4fe(s) + 3 o2(g) < -- --> 2fe2o3(s); which of the followi...
Questions in other subjects:

Biology, 23.07.2019 02:30



Social Studies, 23.07.2019 02:30

Physics, 23.07.2019 02:30

English, 23.07.2019 02:30

Chemistry, 23.07.2019 02:30



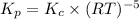

![K_c=\frac{1}{[O_2]^3}\\\\K_c=[O_2]^{-3}](/tpl/images/0067/5962/a0811.png)

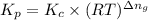
 = number of moles of gaseous products - number of moles of gaseous reactants
= number of moles of gaseous products - number of moles of gaseous reactants

![K_c=[O_2]^{-3}](/tpl/images/0067/5962/00269.png) ,
,  and
and 

