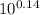Chemistry, 09.07.2019 12:00 kitkattt1246
Suppose you wanted to make a buffer of exactly ph 7.00 using kh2po4 and na2hpo4. if the final solution was 0.1 m in kh2po4, what concentration of na2hpo4 would you need? (pka for h3po4, h2po−4, and hpo2−4 are 2.14, 6.86, and 12.4, respectively.)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:50, adam1299
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments, solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 13:30, amandajbrewerdavis
Table sugar completely dissolved in water is an example of a?
Answers: 1

You know the right answer?
Suppose you wanted to make a buffer of exactly ph 7.00 using kh2po4 and na2hpo4. if the final soluti...
Questions in other subjects:


Arts, 22.01.2021 23:00

Mathematics, 22.01.2021 23:00


Mathematics, 22.01.2021 23:00


Mathematics, 22.01.2021 23:00

Social Studies, 22.01.2021 23:00

Mathematics, 22.01.2021 23:00