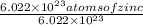Chemistry, 09.07.2019 21:30 ibahadurali786oz4gxs
The enzyme, carbonic anhydrase, is a large zinc-containing protein with a molar mass of 3.00 x10^4 g/mol. zn is 0.218% by mass of the protein. how many zinc atoms does each carbonic anhydrase molecule contain

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:50, nnaomii
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2

Chemistry, 21.06.2019 22:30, kiki197701
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 00:30, thatonestudent2271
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1
You know the right answer?
The enzyme, carbonic anhydrase, is a large zinc-containing protein with a molar mass of 3.00 x10^4 g...
Questions in other subjects:

Chemistry, 02.10.2021 08:20

Mathematics, 02.10.2021 08:20


Chemistry, 02.10.2021 08:20



Business, 02.10.2021 08:20

English, 02.10.2021 08:20


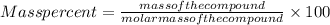
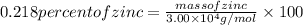
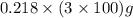
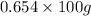 or 65.4 g
or 65.4 g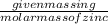
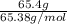
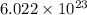 molecules of enzyme consists of
molecules of enzyme consists of