
Chemistry, 10.07.2019 04:00 faithcalhoun
Calculate the mass (in g) of 2.1×1024 atoms of w. calculate the mass (in ) of atoms of . 1.5 ×102g 3.9×102g 2.4 ×102g 6.4 ×102g 3.2 ×102g

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:50, trinityrae4657
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3



Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
Calculate the mass (in g) of 2.1×1024 atoms of w. calculate the mass (in ) of atoms of . 1.5 ×102g 3...
Questions in other subjects:

Computers and Technology, 28.07.2019 22:00







Biology, 28.07.2019 22:00

Health, 28.07.2019 22:00

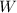 is the symbol of tungsten.
is the symbol of tungsten.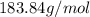
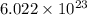
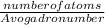
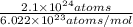
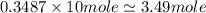
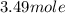 of tungsten consist of
of tungsten consist of 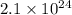 atoms.
atoms.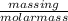
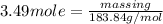
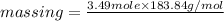
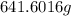 or
or 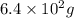
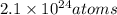 =
= 


