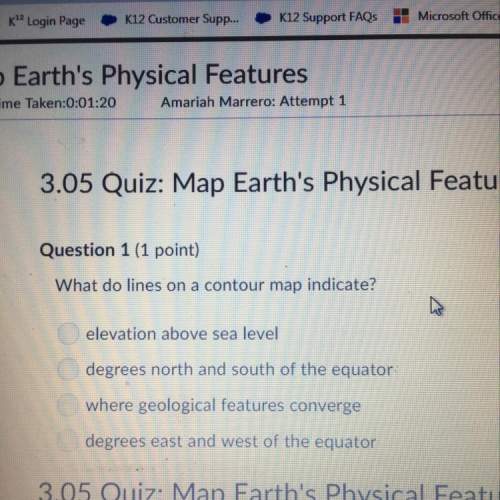
Chemistry, 10.07.2019 05:30 kleathers97
What happens to solubility when the temperature of a solvent is increased.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, kathleendthomas
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 00:50, justabeachbum
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 06:20, stephliu721
What is a property of a double replacement reaction
Answers: 1
You know the right answer?
What happens to solubility when the temperature of a solvent is increased....
Questions in other subjects:


Mathematics, 26.08.2019 11:00

Spanish, 26.08.2019 11:00



History, 26.08.2019 11:10


History, 26.08.2019 11:10