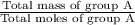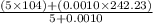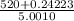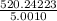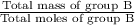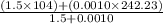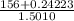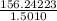Chemistry, 10.07.2019 05:30 elijahjacksonrp6z2o7
Group a used 5.00 moles of styrene and 0.0010 moles of benzoyl peroxide while group b used the same amount of benzoyl peroxide as group a but used only 1.50 moles styrene. what would the average molecular weight of each group's polymer sample be

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:20, phanuel642
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2

Chemistry, 22.06.2019 17:30, mwest200316
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
Group a used 5.00 moles of styrene and 0.0010 moles of benzoyl peroxide while group b used the same...
Questions in other subjects:


Physics, 29.07.2019 01:00




Mathematics, 29.07.2019 01:00