
Chemistry, 10.07.2019 06:00 xbeatdroperzx
Phenol, c6h5oh, is a stronger acid than methanol, ch3oh, even though both contain an o]h bond. draw the structures of the anions resulting from loss of h1 from phenol and methanol, and use resonance structures to explain the difference in acidity.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, laurachealsy923
In an energy pyramid, which level has the most available energy?
Answers: 1



Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1
You know the right answer?
Phenol, c6h5oh, is a stronger acid than methanol, ch3oh, even though both contain an o]h bond. draw...
Questions in other subjects:










 and methanol,
and methanol, 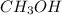 both are alcohols that contain an
both are alcohols that contain an  group attached to carbon atom.
group attached to carbon atom. from phenol, it forms phenoxide anion and due to presence of double bond in the benzene ring the negative charge on the oxygen atom (which represents electrons) will resonate with double bonds of benzene ring as shown in the image. The resonance-stabilized phenoxide ion is more stable. Whereas when methanol lose 1
from phenol, it forms phenoxide anion and due to presence of double bond in the benzene ring the negative charge on the oxygen atom (which represents electrons) will resonate with double bonds of benzene ring as shown in the image. The resonance-stabilized phenoxide ion is more stable. Whereas when methanol lose 1 ![Phenol, c6h5oh, is a stronger acid than methanol, ch3oh, even though both contain an o]h bond. draw](/tpl/images/0072/2100/80d06.jpg)


