

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3


Chemistry, 23.06.2019 05:50, starfox5454
Which of the following isotopes has the same number of neutrons as phosphorus-31?
Answers: 1
You know the right answer?
What is the charge on the tetrapeptide lys-lys-his-glu at ph 7?...
Questions in other subjects:

Biology, 14.11.2019 20:31

Mathematics, 14.11.2019 20:31

Mathematics, 14.11.2019 20:31

Mathematics, 14.11.2019 20:31




History, 14.11.2019 20:31


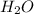 molecule and forming N-terminal(Nitrogen- end) and C-terminal(Carboxyl-end). The bonds formed are known as peptide bonds.
molecule and forming N-terminal(Nitrogen- end) and C-terminal(Carboxyl-end). The bonds formed are known as peptide bonds.
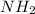 ) groups will be protonated that means they have H-atoms and have (+1) charge.
) groups will be protonated that means they have H-atoms and have (+1) charge.



