Chemistry, 10.07.2019 12:30 hannahpelkey
Solid calcium oxide and gaseous water are formed by the decomposition of solid calcium hydroxide . write a balanced chemical equation for this reaction.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, carter1809
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2


Chemistry, 22.06.2019 12:30, kingbot350
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1
You know the right answer?
Solid calcium oxide and gaseous water are formed by the decomposition of solid calcium hydroxide . w...
Questions in other subjects:





Mathematics, 27.05.2021 04:10




Mathematics, 27.05.2021 04:10

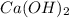 it decomposes to solid calcium oxide CaO and gaseous water
it decomposes to solid calcium oxide CaO and gaseous water 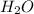 as follows:
as follows: