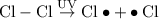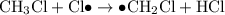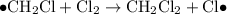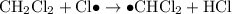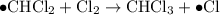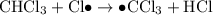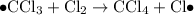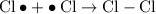Chemistry, 10.07.2019 12:30 bbbbbbbbbbbb62
When light is shown on a mixture of chlorine and chloromethane, carbon tetrachloride is one of the components of the final reaction mixture. propose a series of mechanistic steps which explain this observation?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, livigrace9004
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2


Chemistry, 22.06.2019 14:00, ashlynneboogs0056
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 20:00, denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
When light is shown on a mixture of chlorine and chloromethane, carbon tetrachloride is one of the c...
Questions in other subjects:


Mathematics, 05.12.2020 19:10

Mathematics, 05.12.2020 19:10


Computers and Technology, 05.12.2020 19:10


Mathematics, 05.12.2020 19:10

Mathematics, 05.12.2020 19:10