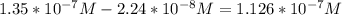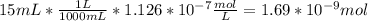Chemistry, 10.07.2019 21:00 deanlmartin
In a typical analysis, 15 ml of an aqueous solution containing an unknown amount of acetylcholine had a ph of 7.65. when incubated with acetylcholinesterase, the ph of the solution decreased to 6.87. assuming there was no buffer in the assay mixture, determine the number of moles of acetylcholine in the 15 ml sample.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 22:20, icantspeakengles
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
In a typical analysis, 15 ml of an aqueous solution containing an unknown amount of acetylcholine ha...
Questions in other subjects:


Mathematics, 27.09.2019 01:30

History, 27.09.2019 01:30



Health, 27.09.2019 01:30




![[H_{3}O^{+}]=10^{-7.65}=2.24*10^{-8} M](/tpl/images/0074/6124/2f1c4.png)
![[H_{3}O^{+}]=10^{-6.87}=1.35*10^{-7} M](/tpl/images/0074/6124/010e5.png)