
Chemistry, 11.07.2019 15:00 wittlemarie
At a certain temperature, the solubility of n2 gas in water at 3.08 atm is 72.5 mg of n2 gas/100 g water . calculate the solubility of n2 gas in water, at the same temperature, if the partial pressure of n2 gas over the solution is increased from 3.08 atm to 8.00 atm . express your answer numerically to three significant figures.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, zamirareece17
1. list three scientific reasons cockroaches may fly.
Answers: 1


Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
At a certain temperature, the solubility of n2 gas in water at 3.08 atm is 72.5 mg of n2 gas/100 g w...
Questions in other subjects:

History, 24.11.2019 13:31

English, 24.11.2019 13:31


Geography, 24.11.2019 13:31


English, 24.11.2019 13:31



Mathematics, 24.11.2019 13:31


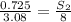
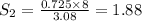
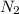 gas in 1 g of water or, 188 mg of tex]N_{2}[/tex] gas in 100 g of water.
gas in 1 g of water or, 188 mg of tex]N_{2}[/tex] gas in 100 g of water.


