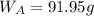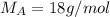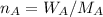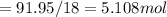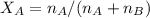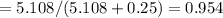A8.05 % ch3oh(aq) has a density of 0.976 g/ml at 18°c what is the mole fraction of the solvent in the solution? 0.953 8.05 91.95 0.0469
2) an aqueous solution of cesium chloride is prepared by dissolving 52.3 g cesium chloride in 60.0g of water at 25°c. the volume of this solution is 63.3 ml . what is the molality of the solution?
0.311 m
4.91 m
2.77 m
5.18 m
3) an aqueous solution of cesium chloride is prepared by dissolving 52.3 g cesium chloride in 60.0g of water at 25°c. the volume of this solution is 63.3 ml . what is the molarity of the solution?
4.91 m
2.69 m
5.18 m
2.77 m
4) the vapor pressure of ethanol, c2h5oh is 100.0 torr at 35 °c. calculate the vapor pressure of the solution formed by dissolving 28.8 g of alpha naphthol, c10h8o, in
36.8 g of c2h5oh. assume alpha naphthol to be nonvolatile at this temperature.
20.0 torr
43.9 torr
80.0 torr
56.1 torr
5) both ethanol, c2h5oh and propanol, c3h7oh, are volatile. at 35 °c, the vapor pressure of pure ethanol is 100 torr and that of propanol is 37.6 torr. what is the vapor pressure at this temperature of a solution is formed by mixing 36.9 g of ethanol and 12.0 g propanol.
15.3 torr
50.1 torr
84.7 torr
87.5 torr
the boiling point of pure ethanol, c2h5oh, is 78.4 latex: ^\circ ∘ c. its boiling point elevation constant is 1.22 °c/m. what is the boiling point of a solution formed by dissolving 8.00 g of alpha-naphthol (c10h7oh) in 100.0 g ethanol.
91.3 degrees centigrade
79.1 degrees centigrade
97.6 degrees centigrade
78.5 degrees centigrade
the freezing point of ccl4 is -22.92°c. calculate the freezing point of the solution prepared by dissolving 17.5g of pyrazine (c4h4n2) in 1250g of ccl4. the freezing point depression constant for ccl4 is 29.8 °c/m.
-22.50
-23.34
-17.71
-28.13
consider the following aqueous solutions:
a. 0.10 m nh4no3,
b. 0.10 m fe(no3)3
c. 0.10 m ba(no3)2
d. 0.10 m nh2conh2
arrange the following in increasing order (smallest to largest) order of osmotic pressure
c < b < a < d
a < d < c < b
d < a < c < b
a < c < b < d
consider the following aqueous solutions:
a. 0.10 m nh4no3,
b. 0.10 m fe(no3)3
c. 0.10 m ba(no3)2
d. 0.10 m nh2conh2
arrange the following in increasing order (smallest to largest) order of freezing point. the freezing point of pure water is 0.00 ∘ c and its freezing point depression constant is 1.86 ∘ c/m
d < a < c < b
b < c < a < d
c < b < a < d
a < c < b < d
consider the following aqueous solutions:
a. 0.10 m nh4no3,
b. 0.10 m fe(no3)3
c. 0.10 m ba(no3)2
d. 0.10 m nh2conh2
arrange the following in increasing order (smallest to largest) order of normal boiling point. the normal boiling point of pure water is 100.00 ∘ c and its boiling point elevation constant is 0.512 ∘ c/m
c < b < a < d
d < a < c < b
a < c < b < d
b < c < a < d
a solution is prepared by dissolving 1.22 g of compound in enough water to make up 262 ml in volume. the osmotic pressure of the solution is found to be 30.3 torr at
35.0 °c. calculate the molar mass of the compound.
257 g/mol
2950 g/mol
3.88 g/mol
44.7 g/mol
i tried to solve them all but i keep getting wrong answers can anyone

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:40, jaueuxsn
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1


Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3
You know the right answer?
A8.05 % ch3oh(aq) has a density of 0.976 g/ml at 18°c what is the mole fraction of the solvent in th...
Questions in other subjects:


Mathematics, 24.11.2019 15:31


Social Studies, 24.11.2019 15:31





Mathematics, 24.11.2019 15:31

Mathematics, 24.11.2019 15:31