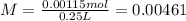Astudent prepares a solution by placing 0.355 g of niso4 · 6 h2o and 0.137 g of nicl2 · 6 h2o in a 250-ml volumetric flask. a small amount of distilled water is then added, and the compounds are swirled in the flask until they dissolve. after they have dissolved, the solution is diluted to the mark. what is the molar concentration of cl− ions in the final solution? answer in units of m.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, rosieposie27
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1



Chemistry, 23.06.2019 00:00, savyblue1724707
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
Astudent prepares a solution by placing 0.355 g of niso4 · 6 h2o and 0.137 g of nicl2 · 6 h2o in a 2...
Questions in other subjects:



Mathematics, 28.04.2021 18:10



Spanish, 28.04.2021 18:10


Mathematics, 28.04.2021 18:10