
There are two binary compounds of mercury and oxygen. heating either of them results in the decomposition of the compound, with oxygen gas escaping into the atmosphere while leaving a residue of pure mercury. heating 0.6498 g of one of the compounds leaves a residue of 0.6018 g. heating 0.4172 g of the other compound results in a mass loss of 0.016 g. determine the empirical formula of each compound.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:10, Kianna000
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 12:30, americanbellabeauty
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1


Chemistry, 22.06.2019 17:10, mikeeway33
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2
You know the right answer?
There are two binary compounds of mercury and oxygen. heating either of them results in the decompos...
Questions in other subjects:

History, 03.02.2021 22:50

Mathematics, 03.02.2021 22:50




Mathematics, 03.02.2021 22:50

Arts, 03.02.2021 22:50

Mathematics, 03.02.2021 22:50


Chemistry, 03.02.2021 22:50

 and
and 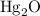 .
.
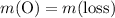
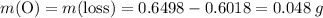
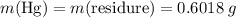
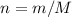 ;
; 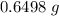 of the first compound would contain
of the first compound would contain 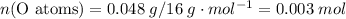
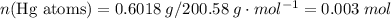
 ratio; thus the empirical formula for this compound would be
ratio; thus the empirical formula for this compound would be 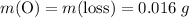
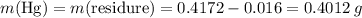
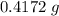 of the first compound would contain
of the first compound would contain 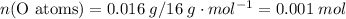
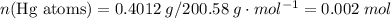
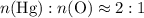 and therefore the empirical formula
and therefore the empirical formula

