

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:40, taysomoneyyy
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 09:20, kevinhernandez582
What will most likely happen when two bromine atoms bond together?
Answers: 3


Chemistry, 22.06.2019 14:30, Playboycxm
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1
You know the right answer?
Titanium (ti) has an hcp crystal structure, a density of 4.51 g/cm3, and the atomic weight for ti, a...
Questions in other subjects:



English, 20.02.2020 09:59

English, 20.02.2020 10:00





Mathematics, 20.02.2020 10:01

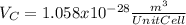

 :
: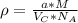
 for the Avogadro number, thus:
for the Avogadro number, thus:

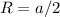 and
and  , thus:
, thus:![V_C=6c(a/2)^2\sqrt{3}\\V_C=6a*1.58(a/2)^2\sqrt{3}\\V_C=16.4a^3/4\\a=\sqrt[3]{\frac{4*1.058x10^{-28}\frac{m^3}{UnitCell}}{16.4} } \\a=2.96x10^{-10}m\\c=1.58a\\c=1.58*2.96x10^{-10}m\\c=4.67x10^{-10}m](/tpl/images/0081/7540/a224e.png)



