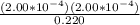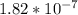Chemistry, 13.07.2019 21:30 ivanyeli4520
The equilibrium concentrations of the reactants and products are [ha] = 0.220 m [h3o ] = 2.00 × 10–4 m [a–] = 2.00 × 10–4 m calculate the ka value for the acid ha.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:50, Jerrikasmith28
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 21:50, BookandScienceNerd
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 23.06.2019 00:00, juliannasl
How is the way a mixture is combined different from how a compound is combined?
Answers: 3
You know the right answer?
The equilibrium concentrations of the reactants and products are [ha] = 0.220 m [h3o ] = 2.00 × 10–4...
Questions in other subjects:

Mathematics, 06.04.2021 17:10



Social Studies, 06.04.2021 17:10

Mathematics, 06.04.2021 17:10

Biology, 06.04.2021 17:10

History, 06.04.2021 17:10

Physics, 06.04.2021 17:10

Biology, 06.04.2021 17:10

Mathematics, 06.04.2021 17:10

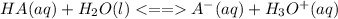
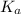 is the equilibrium constant for this equation, which is referred to as the acid dissociation constant.
is the equilibrium constant for this equation, which is referred to as the acid dissociation constant. ![K_{a} = \frac{[H_{3}O^{+}][A^{-}]}{[HA]}](/tpl/images/0086/2201/14aa4.png)