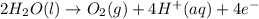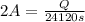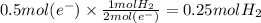Chemistry, 14.07.2019 10:00 nekathadon
In the electrolysis of water shown below, a current of 2 amps is applied to 180 ml of h2o(l) for 6 hours and 42 minutes. how many grams of h2(g) are formed? (faraday's constant = 96,500 c/mol)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:10, kellinvagneur
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1

Chemistry, 22.06.2019 23:30, adamgala3885
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 00:00, tonimgreen17p6vqjq
The graph indicates the running route for tobias. which best describes his run? from time 0 to 6, he went fast and then slowed down. from time 6 to 10, he was at his slowest. from time 12 to 14, he went very slow. from time 14 to 18, he went toward the starting point.
Answers: 2
You know the right answer?
In the electrolysis of water shown below, a current of 2 amps is applied to 180 ml of h2o(l) for 6 h...
Questions in other subjects:





Mathematics, 04.01.2020 03:31



Biology, 04.01.2020 03:31

History, 04.01.2020 03:31