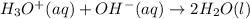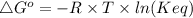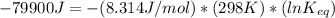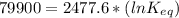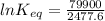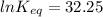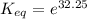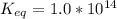Chemistry, 14.07.2019 10:30 samantha636
The standard free energy change for the formation of two moles of h2o(l) in a strong acid–strong base neutralization reaction at 25°c is -79.9kj. calculate the equilibrium constant for the reaction. see equation 11.1. h3o+(aq) + oh-(aq) = 2 h2o (l)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 14:30, belindajolete
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2


Chemistry, 22.06.2019 18:00, darrell1168
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
The standard free energy change for the formation of two moles of h2o(l) in a strong acid–strong bas...
Questions in other subjects:

Biology, 16.12.2020 01:00

Mathematics, 16.12.2020 01:00





English, 16.12.2020 01:00

Mathematics, 16.12.2020 01:00


Mathematics, 16.12.2020 01:00