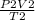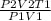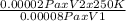Chemistry, 14.07.2019 11:00 bryanmcmillianjr
Agas at 250 k has a pressure of 0.00008 pa. if the pressure drops to 0.00002 pa, what is the new temperature? 500 k 1000 k 125 k 62.5 k

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2


Chemistry, 22.06.2019 20:00, denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
Agas at 250 k has a pressure of 0.00008 pa. if the pressure drops to 0.00002 pa, what is the new tem...
Questions in other subjects:


Social Studies, 19.11.2020 20:20




Mathematics, 19.11.2020 20:20


Mathematics, 19.11.2020 20:20


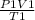 =
=