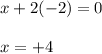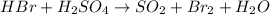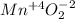Chemistry, 14.07.2019 23:30 carlosbs71
1) assign oxidation numbers to each element in this compound. mno₂ mn = o = 2) hbr + h2so4 ↔ so2 + br2 + h2o oxidizing agent = reducing agent =

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, foreignking02
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 13:30, makenziehook8
Which is true of a liquid? it has a definite volume but not a definite mass. it has a definite mass but not a definite volume. it has a definite volume but not a definite shape. it has a definite shape but not a definite volume.
Answers: 2


Chemistry, 23.06.2019 04:31, CassidgTab
Which molecules are more strongly attracted to one another -c3h8o molecules that make up liquid rubbing alcohol or ch4 molecules that make up methane gas
Answers: 3
You know the right answer?
1) assign oxidation numbers to each element in this compound. mno₂ mn = o = 2) hbr + h2so4 ↔ so2 +...
Questions in other subjects:


Mathematics, 13.09.2019 02:30

English, 13.09.2019 02:30

Social Studies, 13.09.2019 02:30





History, 13.09.2019 02:30

Mathematics, 13.09.2019 02:30

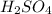 and reducing agent is HBr.
and reducing agent is HBr.