Chemistry, 15.07.2019 03:00 hdhdenjdxk
What volume of a 2.75 m solution of naoh is required to make 500.0 ml of a 1.27 m solution of naoh?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:50, lildestinyquintana
Ase your answer to this question on the information below. hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant. the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen. chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product. nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 08:30, audrey1256
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 14:00, ashlynneboogs0056
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3
You know the right answer?
What volume of a 2.75 m solution of naoh is required to make 500.0 ml of a 1.27 m solution of naoh?...
Questions in other subjects:

Mathematics, 26.08.2020 21:01

Computers and Technology, 26.08.2020 21:01

Mathematics, 26.08.2020 21:01

Business, 26.08.2020 21:01

English, 26.08.2020 21:01


Mathematics, 26.08.2020 21:01


Mathematics, 26.08.2020 21:01

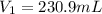
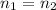
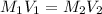
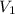 as it is the volume we need to take from the inital 2.75M solution of NaOH, thus:
as it is the volume we need to take from the inital 2.75M solution of NaOH, thus: