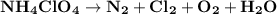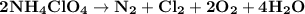Chemistry, 15.07.2019 10:00 angelaencinas90
Ammonium perchlorate nh4clo4 is a powerful solid rocket fuel, used in the space shuttle boosters. it decomposes into nitrogen n2 gas, chlorine cl2 gas, oxygen o2 gas and water vapor, releasing a great deal of energy. calculate the moles of ammonium perchlorate needed to produce 0.10mol of chlorine. be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:20, payshencec21
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1



Chemistry, 23.06.2019 04:00, Tiredd7838
Which of these are physical changes in matter? check all that apply boiling water a pencil being sharpened exploding dynamite freezing water rotting cheese
Answers: 1
You know the right answer?
Ammonium perchlorate nh4clo4 is a powerful solid rocket fuel, used in the space shuttle boosters. it...
Questions in other subjects:

Advanced Placement (AP), 03.01.2020 06:31







Mathematics, 03.01.2020 06:31


Physics, 03.01.2020 06:31