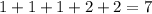Chemistry, 17.07.2019 12:30 SKYBLUE1015
Given the unbalanced equation: (oh)2 + )2so4 → + + what is the sum of the coefficients when the equation is completely balanced using the smallest whole-number coefficients?

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 21:30, starl0rd211
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2
You know the right answer?
Given the unbalanced equation: (oh)2 + )2so4 → + + what is the sum of the coefficients when the...
Questions in other subjects:





Mathematics, 26.03.2021 18:30



Mathematics, 26.03.2021 18:30

Mathematics, 26.03.2021 18:30

Mathematics, 26.03.2021 18:30