
Chemistry, 17.07.2019 18:00 andybiersack154
The gas left in a used aerosol can is at a pressure of 103 kpa at 25.0 °c. if the can heats up to 50.0 °c, what is the pressure of the gas inside the can, assuming the volume of the can is constant?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:40, jude3412
In an effort to address concerns about global warming, a power plant in portland, oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

You know the right answer?
The gas left in a used aerosol can is at a pressure of 103 kpa at 25.0 °c. if the can heats up to 50...
Questions in other subjects:




English, 16.04.2020 04:38



Social Studies, 16.04.2020 04:38



Mathematics, 16.04.2020 04:38

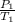 =
= 


