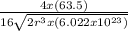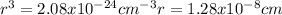Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, JuniperGalaxy
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1



Chemistry, 23.06.2019 13:50, isaac7454
Use the periodic table and your knowledge of isotopes to complete these statements. when polonium-210 emits an alpha particle, the child isotope has an atomic mass of 1-131 undergoes beta-minus decay. the chemical symbol for the new element is fluorine-18 undergoes beta-plus decay. the child isotope has an atomic mass of done intro donne
Answers: 1
You know the right answer?
Copper has a face-centered cubic unit cell. the density of copper is 8.96 g/cm3. calculate a value f...
Questions in other subjects:

Mathematics, 16.07.2021 22:30



Mathematics, 16.07.2021 22:30

Mathematics, 16.07.2021 22:30

Mathematics, 16.07.2021 22:30


Mathematics, 16.07.2021 22:30


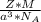
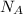 is Avogadro's number, and a is the edge length.
is Avogadro's number, and a is the edge length.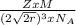
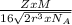
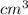 =
=