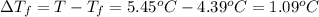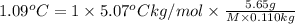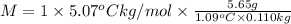Chemistry, 20.07.2019 15:30 emmanuelmashao5504
Asolution is made by dissolving 5.65 g of an unknown molecular compound in 110.0 g of benzene froze at 4.39 oc. what is the molar mass of the solute if pure benzene has a freezing point of 5.45 oc and the kf value of benzene is 5.07 oc/m

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 23.06.2019 12:30, hollycoleman13
Choose one literary selection from this semester in which you think the setting has a great impact on the work. in a full paragraph name the work, describe the setting, and explain why it is so important to the overall story or poem.
Answers: 1

Chemistry, 23.06.2019 15:00, maariaaa10
An isotope undergoes radioactive decay by emitting radiation that has no mass. what other characteristic does the radiation have?
Answers: 3
You know the right answer?
Asolution is made by dissolving 5.65 g of an unknown molecular compound in 110.0 g of benzene froze...
Questions in other subjects:

Mathematics, 13.07.2019 14:00

English, 13.07.2019 14:00


English, 13.07.2019 14:00


Mathematics, 13.07.2019 14:00


Advanced Placement (AP), 13.07.2019 14:00


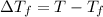
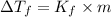
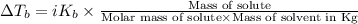
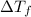 =Depression in freezing point
=Depression in freezing point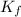 = freezing point constant of solvent= 5.07°C/m
= freezing point constant of solvent= 5.07°C/m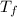 =4.39 °C
=4.39 °C