
Chemistry, 20.07.2019 20:00 xxYingxYangxx7311
Asolution is made by dissolving 24 g of nacl to make 475 ml of solution. calculate the concentration in units of molarity by following these steps: a) convert the grams of nacl to moles of nacl. b) calculate the liters of solution by dividing the given milliliters by 1000. c) divide moles by the liters of solution.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, njones58emailtjcedu
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 15:30, neariah24
Plz me ! 1 which of earths spheres contains most of its mass? a atmosphere b hydrosphere c geosphere* d biosphere 2 erosion and weathering are examples of which types of forces? a constructive forces b destructive forces* c gravitational forces d inertia-related forces 3 which of the following statements about earths atmosphere is true? a earths atmosphere contains 78% water vapor which is essentail to life b earths atmosphere contains 21% oxygen c earths atmosphere contains carbon dioxide which all life forms require d earths atmosphere allows radiation from the sun to pass through it and warm earths surface* 4 the strenght of the force of gravity between two objects is determined by which of the following factors? select all that apply a the messes of the objects* b the distance between the objects* c the volumes of the objects d the surface area of the objects 5 earth and moon are kept in there respective orbits due to the influence of a inertia b gravity c gravity and inertia* d neither gravity or inertia if you answer all questions right i will give
Answers: 1

Chemistry, 22.06.2019 19:30, youngdelvin123
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Asolution is made by dissolving 24 g of nacl to make 475 ml of solution. calculate the concentration...
Questions in other subjects:


Mathematics, 25.03.2020 08:56


Chemistry, 25.03.2020 08:56

Computers and Technology, 25.03.2020 08:56



Mathematics, 25.03.2020 08:56


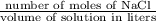 =
= 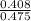 = 0.8589 M
= 0.8589 M


