
Chemistry, 20.07.2019 21:30 leeahnnfoster
Butane c4 h10 (-1.jpghf = –125.7), combusts in the presence of oxygen to form co2 (g) (mc016-2.jpghf = –393.5 kj/mol), and h2 o(g) (mc016-3.jpghf = –241.82) in the reaction: mc016-4.jpg what is the enthalpy of combustion, per mole, of butane?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, rosetoheart2
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2



You know the right answer?
Butane c4 h10 (-1.jpghf = –125.7), combusts in the presence of oxygen to form co2 (g) (mc016-2.jpghf...
Questions in other subjects:


Geography, 12.03.2021 22:50




Biology, 12.03.2021 22:50



Chemistry, 12.03.2021 22:50

Biology, 12.03.2021 22:50

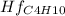 = -125.7 kJ/mol
= -125.7 kJ/mol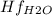 = -241.82 kJ/mol
= -241.82 kJ/mol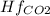 = -393.5 kJ/mol
= -393.5 kJ/mol

