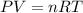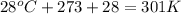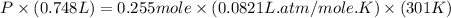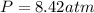Chemistry, 21.07.2019 04:00 robertstoll81
Question 1 a sample of 0.255 mole of gas has a volume of 748 ml at 28°c. calculate the pressure of this gas. (r= 0.0821 l ∙ atm / mol ∙ k) 0.784 atm 8.42 atm 0.00842 atm 7.84 × 10-4 atm none of the above

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:30, lileljusto2829
Which is the most likely way an automotive engineer would use chemistry
Answers: 1


Chemistry, 22.06.2019 13:30, justinerodriguz2878
What are the major types of a chemical compound
Answers: 2
You know the right answer?
Question 1 a sample of 0.255 mole of gas has a volume of 748 ml at 28°c. calculate the pressure of t...
Questions in other subjects: