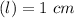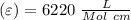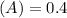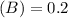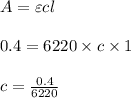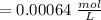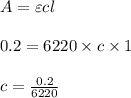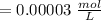Using and spectrophotometer, you measure 2 dilutions of NADH, and get absorbance values of 0.4 for sample A, and 0.2 for sample B. You know that the path length is 1 cm, and the extinction coefficient for NADH is 6220 (L Morcm). Using the Lambert-Beer Law equation (below), calculate the concentrations of sample A Select] and Sample B (Select ] A = log10 () = Ecl Where: A- Absorbance C- Concentration (mol 1 - Path length (cm) E = molar decadic extinction coefficient L mol. cm 1o - Intensity of the incident light 1 - Intensity of the transmitted Night

Answers: 3


Other questions on the subject: Biology


Biology, 22.06.2019 04:00, shayshayyy41
What is the difference between how ionic and covalent bonds form
Answers: 1


Biology, 22.06.2019 11:00, ayoismeisalex
Which of these is true of the cytoplasm of an unfertilized egg? a. it is an unevenly distributed mixture of mrna, proteins, organelles, and other substances. b. it does not contain substances that are important in directing development. c. these substances are supplied by the sperm. d. it does not contain substances that are important in directing development. e. development is directed solely by the surrounding cells. f. it is a homogeneous mixture of mrna, proteins, organelles, and other substances. g. it does not contain substances that are important in directing development. h. these substances are produced by the dna of the fertilized zygote.
Answers: 1
You know the right answer?
Using and spectrophotometer, you measure 2 dilutions of NADH, and get absorbance values of 0.4 for s...
Questions in other subjects:


Social Studies, 13.12.2019 00:31








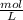 and concentration of sample B is 0.00003
and concentration of sample B is 0.00003