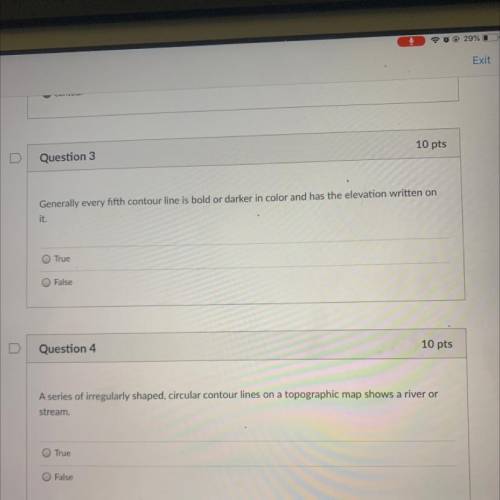
What is the answer to number three and four I’m like totally confused
...

Biology, 29.04.2021 22:30 cdyshaylia55
What is the answer to number three and four I’m like totally confused

Answers: 2


Other questions on the subject: Biology

Biology, 22.06.2019 03:00, historyfanboy101
Wich organism obtains its energy from dead or decaying matter
Answers: 1

Biology, 22.06.2019 06:00, tiffanibell71
Which kingdom includes some organisms that have no nucleus and can live in an environment with an extremely high salt content
Answers: 1

Biology, 22.06.2019 07:00, doris8051
What was the purpose of mendel's experiments with dihybrid crosses? a. to determine if dna was a transforming factor b. to determine if traits could be recessive c. to determine if traits affected each other d. to determine if traits had more than one allele
Answers: 3

Biology, 22.06.2019 08:40, keilyjaramillo2870
What best explains whether bromine (br) or neon (ne) is more likely to form a covalent bond? bromine forms covalent bonds because it has seven valence electrons, but neon has eight valence electrons and already fulfills the octet rule. bromine forms covalent bonds because it has many electron shells, but neon has only two electron shells and is tightly bound to its electrons. neon forms covalent bonds because it can share its valence electrons, but bromine has seven valence electrons and can gain only one more electron. neon forms covalent bonds because it has only two electron shells, but bromine has many electron shells and will lose electrons in order to fulfill the octet rule.
Answers: 3
You know the right answer?
Questions in other subjects:

Mathematics, 04.01.2021 20:50

Arts, 04.01.2021 20:50

Social Studies, 04.01.2021 20:50

Medicine, 04.01.2021 20:50

Mathematics, 04.01.2021 20:50




History, 04.01.2021 20:50

Computers and Technology, 04.01.2021 20:50



