
Biology, 27.10.2020 17:40 kiarajack456
Can someone please answer this because I have no clue and I’ve been waiting two hours for an answer
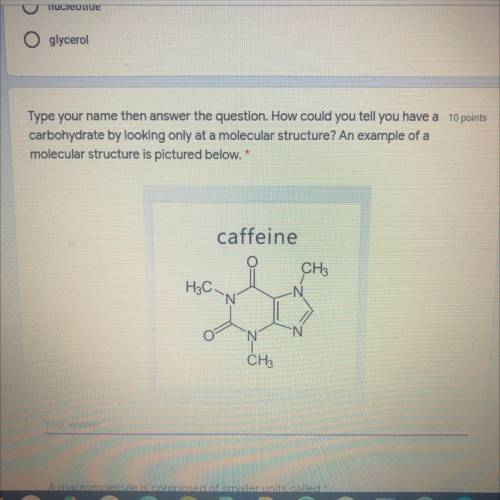

Answers: 1


Other questions on the subject: Biology

Biology, 22.06.2019 08:40, keilyjaramillo2870
What best explains whether bromine (br) or neon (ne) is more likely to form a covalent bond? bromine forms covalent bonds because it has seven valence electrons, but neon has eight valence electrons and already fulfills the octet rule. bromine forms covalent bonds because it has many electron shells, but neon has only two electron shells and is tightly bound to its electrons. neon forms covalent bonds because it can share its valence electrons, but bromine has seven valence electrons and can gain only one more electron. neon forms covalent bonds because it has only two electron shells, but bromine has many electron shells and will lose electrons in order to fulfill the octet rule.
Answers: 3


Biology, 23.06.2019 00:20, emalvidrez5205
Availability of food: a. is dependent upon kin selection. b. can be highly variable, depending on season and rainfall. c. is determined by the alpha male only. d. is restricted to individuals in the dominant hierarchy.
Answers: 1
You know the right answer?
Can someone please answer this because I have no clue and I’ve been waiting two hours for an answer...
Questions in other subjects:

Mathematics, 05.05.2020 23:39

Mathematics, 05.05.2020 23:39


Mathematics, 05.05.2020 23:39

Biology, 05.05.2020 23:39



English, 05.05.2020 23:39

Mathematics, 05.05.2020 23:39



